| |
p53 Information
mdm family
mouse models
ASPP family
|
p53 in development
A good review dealing with the possibility of an important role of p53 as a protector during development
Hall, P. A. and D. P. Lane (1997). “Tumour suppressors: A developing role for p53?” Curr Biol 7: R144-R147.
p53 Knockout mice phenotype
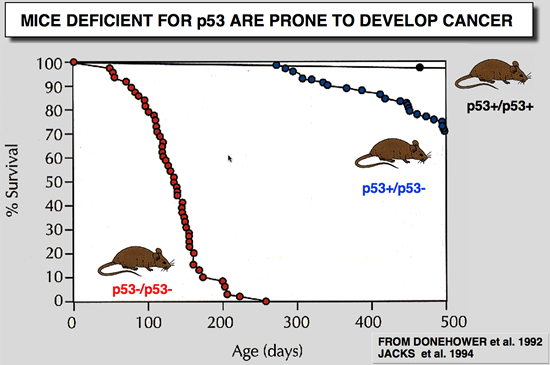 |
Donehower LA, Harvey M, Slagle BL, Mcarthur MJ, Montgomery CA, Butel JS and Bradley A (1992) Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356: 215-221.
Abstract of the paper
Mutations in the p53 tumour-suppressor gene are the most frequently observed genetic lesions in human cancers. To investigate the role of the p53 gene in mammalian development and tumorigenesis, a null mutation was introduced into the gene by homologous recombination in murine embryonic stem cells. Mice homozygous for the null allele appear normal but are prone to the spontaneous development of a variety of neoplasms by 6 months of age. These observations indicate that a normal p53 gene is dispensable for embryonic development, that its absence predisposes the animal to neoplastic disease, and that an oncogenic mutant form of p53 is not obligatory for the genesis of many types of tumours |
p53 Knockout mice phenotype: Effects of genetic background
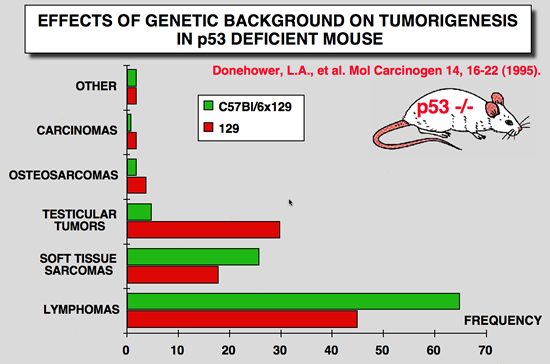 |
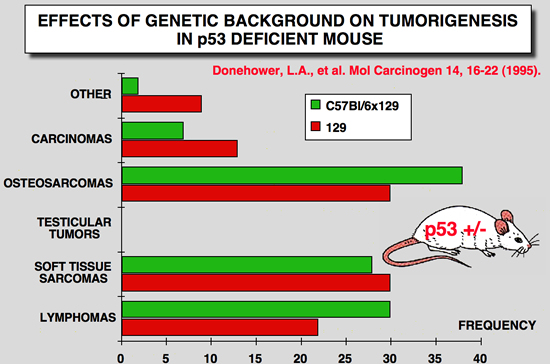 |
Donehower LA, Harvey M, Vogel H, McArthur MJ, Montgomery CA, Jr., Park SH, Thompson T, Ford RJ and Bradley A (1995) Effects of genetic background on tumorigenesis in p53-deficient mice. Mol Carcinog 14: 16-22.
Abstract of the paper
Mice with disrupted germline p53 alleles have been engineered by us and others and have been shown to have enhanced susceptibility to spontaneous tumors of various types. We monitored a large number of p53- deficient mice (p53+/- and p53-/-) and their wild-type littermates (p53+/+) of two different genetic backgrounds (129/Sv and mixed C57BL/6 x 129/Sv) up to 2 yr of age. p53+/- and p53-/- 129/Sv mice show accelerated tumorigenesis rates compared with their p53-deficient counterparts of mixed C57BL/6 x 129/Sv genetic background. The tumor spectra of the two strains of mice are similar except that almost half of 129/Sv p53-/- males develop malignant teratomas, whereas these tumors are rarely observed in C57BL/6 x 129/Sv mice and never in 129/Sv p53+/- males. In the study reported here, we further characterized the lymphomas that arose in the p53-nullizygous mice and found that over three-quarters of the lymphomas were of thymic origin and contained primarily immature (CD4+/CD8+) T-cells, whereas the remainder originated in the spleen and peripheral lymph nodes and were of B-cell type. The high incidence of early-onset lymphomas in the nullizygous mice makes these animals a good lymphoma model, whereas the heterozygous mice may be a useful model for Li-Fraumeni syndrome, a human inherited cancer predisposition |
p53 Knockout mice phenotype: developmental abnormalities
 |
Rotter V, Schwartz D, Almon E, Goldfinger N, Kapon A, Meshorer A, Donehower LA and Levine AJ (1993) Mice with reduced levels of p53 protein exhibit the testicular giant- cell degenerative syndrome. Proc Natl Acad Sci U S A 90: 9075-9.
Abstract of the paper
Defects in neural tube formation are among the most common malformations leading to infant mortality. Although numerous genetic loci appear to contribute to the defects observed in humans and in animal model systems, few of the genes involved have been characterized at the molecular level. Mice lacking the p53 tumour suppressor gene are predisposed to tumours, but the viability of these animals indicates that p53 function is not essential for embryonic development. Here, we demonstrate that a fraction of p53-deficient embryos in fact do not develop normally. These animals display defects in neural tube closure resulting in an overgrowth of neural tissue in the region of the mid- brain, a condition known as exencephaly
|
Armstrong JF, Kaufman MH, Harrison DJ and Clarke AR (1995) High-frequency developmental abnormalities in p53-deficient mice. Curr Biol 5: 931-6.
Abstract of the paper
BACKGROUND: Several strains of mice carrying null mutations of the tumour suppressor gene p53 have been developed. It has been reported that homozygous mice from all of these strains develop normally to birth, but then succumb rapidly to neoplasia. RESULTS: Here, we report that a significant proportion of female p53-/- mice die during embryogenesis or in the period between birth and weaning, being subject to a spectrum of abnormalities. In a significant proportion (23%) of p53-/- female embryos, the normal process of neural tube closure failed, leading to exencephaly and subsequent anencephaly. Although this phenomenon was predominantly associated with females, we observed one affected male embryo. In addition to a spectrum of neural tube defects, many of these embryos exhibited a range of craniofacial malformations, including ocular abnormalities and defects in upper incisor tooth formation. We observed a significant reduction in the number of p53-/- female progeny of p53+/- x p53+/- matings, and also in an in utero analysis of the p53+/- female progeny of p53-/- x p53+/+ matings. When male mice were exposed to irradiation prior to mating, a significant increase in the rate of abnormality was seen in the progeny, which was specifically associated with p53 deficiency. CONCLUSIONS: We have identified a high rate of developmental abnormalities associated with p53 deficiency. This manifests itself as a spectrum of lesions, predominantly female-associated defects in neural tube closure. These defects may arise either because p53 plays a physiological role at the time of neural tube closure, or because of an abnormally high frequency of mutation within the haploid gametes of p53- null parents.
|
Sah VP, Attardi LD, Mulligan GJ, Williams BO, Bronson RT and Jacks T (1995) A subset of p53-deficient embryos exhibit exencephaly. Nat Genet 10: 175-80.
Abstract of the paper
Transgenic mice which carry hybrid p53 promoter-chloramphenicol acetyltransferase (CAT) transgenes were found to express CAT enzymatic activity predominantly in the testes. Endogenous levels of p53 mRNA and protein were lower than in the nontransgenic control mice. The various p53 promoter-CAT transgenic mice exhibited in their testes multinucleated giant cells, a degenerative syndrome resulting presumably from the inability of the tetraploid primary spermatocytes to complete meiotic division. The giant-cell degenerative syndrome was also observed in some genetic strains of homozygous p53 null mice. In view of the hypothesis that p53 plays a role in DNA repair mechanisms, it is tempting to speculate that the physiological function of p53 that is specifically expressed in the meiotic pachytene phase of spermatogenesis is to allow adequate time for the DNA reshuffling and repair events which occur at this phase to be properly completed. Primary spermatocytes which have reduced p53 levels are probably impaired with respect to DNA repair, thus leading to the development of genetically defective giant cells that do not mature
|
p53 Knockout mice phenotype: susceptibility to teratogenicity
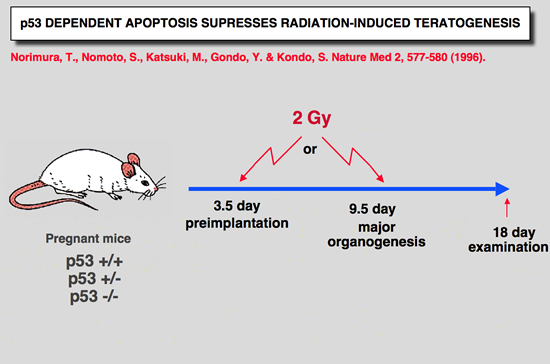 |
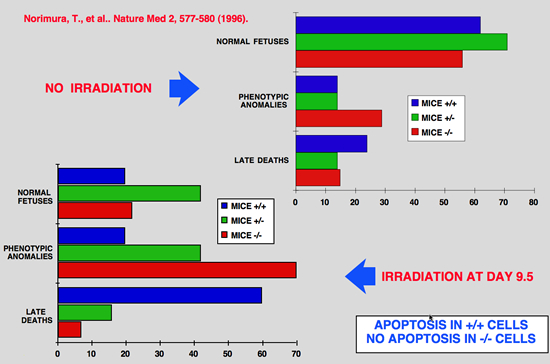 |
Norimura T, Nomoto S, Katsuki M, Gondo Y and Kondo S (1996) p53-dependent apoptosis suppresses radiation-induced teratogenesis. Nature Med 2: 577-580.
Abstract of the paper
About half of human conceptions are estimated not to be implanted in the uterus, resulting in unrecognizable spontaneous abortions(1,2), and about 5% of human births have a recognizable malformation(1,3). In order to find clues to the mechanisms of malformation and abortion, we compared the incidences of radiation-induced malformations and abortions in p53 null (p53(-/-)) and wild-type (p53(+/+)) mice. After X-irradiation with 2 Cy on day 9.5 of gestation, p53(-/-) mice showed a 70% incidence of anomalies and a 7% incidence of deaths, whereas p53(+/+) mice had a 20% incidence of anomalies and a 60% incidence of deaths. Similar results were obtained after irradiation on day 3.5 of gestation. This reciprocal relationship of radiosensitivity to anomalies and to embryonic or fetal lethality supports the notion that embryonic or fetal tissues have a p53-dependent ''guardian'' of the tissue(4) that aborts cells bearing radiation-induced teratogenic DNA damage. In fact, after X-irradiation, the number of cells with apoptotic DNA fragments was greatly increased in tissues of the p53(+/+) fetuses but not in those of the p53(-/-) fetuses. |
 |
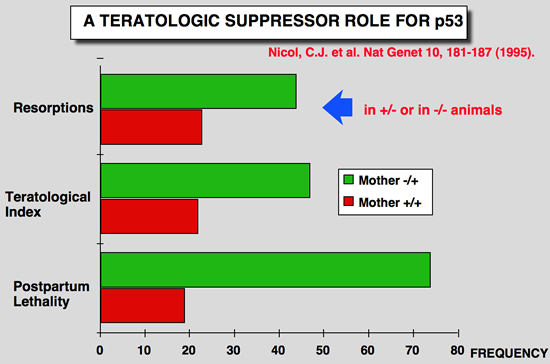 |
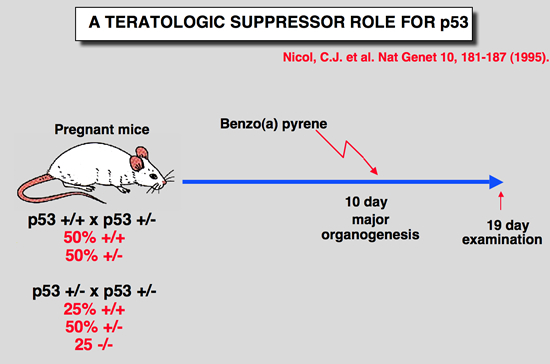
Nicol CJ, Harrison ML, Laposa RR, Gimelshtein IL and Wells PG (1995) A teratologic suppressor role for p53 in benzo[a]pyrene-treated transgenic p53-deficient mice. Nat Genet 10: 181-187.
Abstract of the paper
DNA damage may mediate birth defects caused by many drugs and environmental chemicals, therefore p53, a tumour suppressor gene that facilitates DNA repair, may be critically embryoprotective. We have studied the effects of the environmental teratogen, benzo[a]pyrene, on pregnant heterozygous p53-deficient mice. Such mice exhibited between 2- to 4-fold higher embryotoxicity and teratogenicity than normal p53- controls. Fetal resorptions reflecting in utero death were genotyped using the polymerase chain reaction and found to be increased 2.6-fold and 3.6-fold respectively with heterozygous and homozygous p53- deficient embryos. These results provide the first direct evidence that p53 may be an important teratological suppressor gene which protects the embryo from DNA-damaging chemicals and developmental oxidative stress. |
 |
| Brash DE (1996) Cellular proofreading. Nature Med. 2: 525-526. |
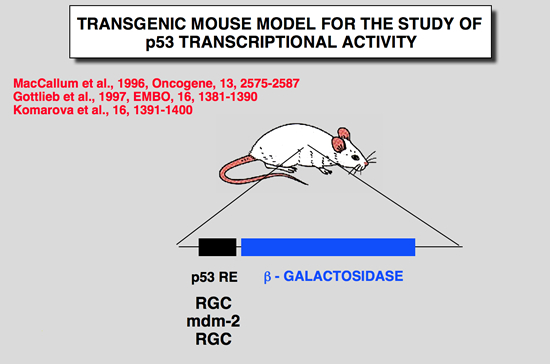
p53 mice models for early development study
 |
 |
 |
 |
 |
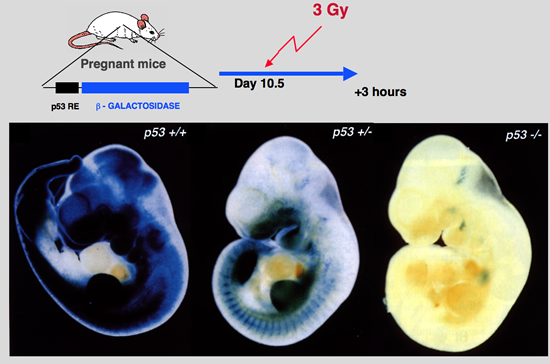
Komarova, E. A., M. V. Chernov, et al. (1997). “Transgenic mice with p53-responsive lacZ: P53 activity varies dramatically during normal development and determines radiation and drug sensitivity in vivo.” Embo J 16: 1391-1400.
Abstract of the paper
To analyze the involvement of p53-dependent transcriptional activation in normal development and in response to DNA damage in vivo, we created transgenic mice with a lacZ reporter gene under the control of a p53- responsive promoter. Five independent strains showed similar patterns of transgene expression. In untreated animals, lacZ expression was limited to the developing nervous system of embryos and newborn mice and was strongly decreased in the adult brain. Gamma-irradiation or adriamycin treatment induced lacZ expression in the majority of cells of early embryos and in the spleen, thymus and small intestine in adult mice. Transgene expression was p53 dependent and coincided with the sites of strong p53 accumulation. The lacZ-expressing tissues and early embryos, unlike other adult tissues and late embryos, are characterized by high levels of p53 mRNA expression and respond to DNA damage by massive apoptotic cell death, Analysis of p53-null mice showed that this apoptosis is p53 dependent. These data suggest that p53 activity, monitored by the reporter lacZ transgene, is the determinant of radiation and drug sensitivity in vivo and indicate the importance of tissue and stage specificity of p53 regulation at the level of mRNA expression. |
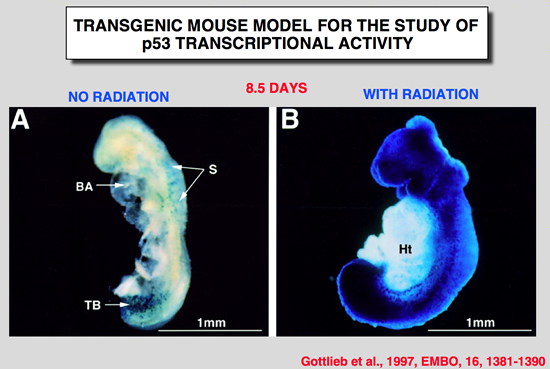
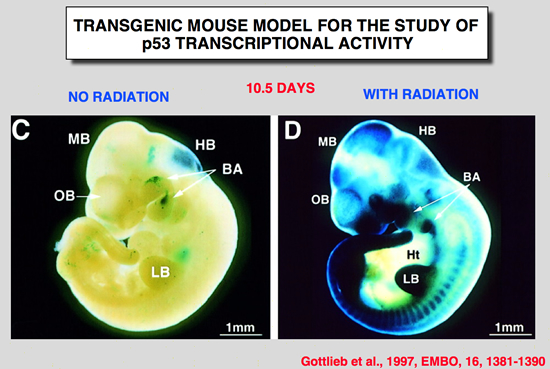
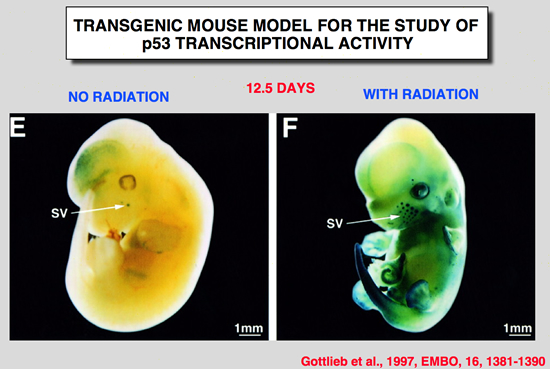
Gottlieb, E., R. Haffner, A. King, G. Asher, P. Gruss, P. Lonai and M. Oren (1997). “Transgenic mouse model for studying the transcriptional activity of the p53 protein: Age- and tissue-dependent changes in radiation-induced activation during embryogenesis.” Embo J 16: 1381-1390.
Abstract of the paper
The p53 tumor suppressor protein is a sequence-specific transcriptional activator of target genes, Exposure of cells to DNA damage results in accumulation of biochemically active p53, with consequent activation of p53-responsive promoters, In order to study how the transcriptional activity of the p53 protein is regulated in vivo, a transgenic mouse strain was generated, These mice harbor the p53-dependent promoter of the mdm2 gene, fused to a lacZ reporter gene. Induction of lacZ activity by DNA damage (ionizing radiation) was monitored in embryos of different p53 genotypes, The transgenic promoter was substantially activated in vivo following irradiation; activation required functional p53, The activation pattern became more restricted with increasing embryo age, as well as with the state of differentiation of a given tissue, Generally, maximal p53 activation occurred in rapidly proliferating, relatively less differentiated cells, A striking extent of haploinsufficiency was revealed-induction of promoter activity was far less efficient in mice carrying only one wild-type p53 allele, This suggests that normal levels of cellular p53 are limiting, and any further reduction already compromises the p53 response significantly, Thus, the activation potential of p53 is tightly controlled in vivo, both spatially and temporally, and an important element in this control is the presence of limiting basal levels of activatable p53. |
Heterogeneity of p53 response in vivo
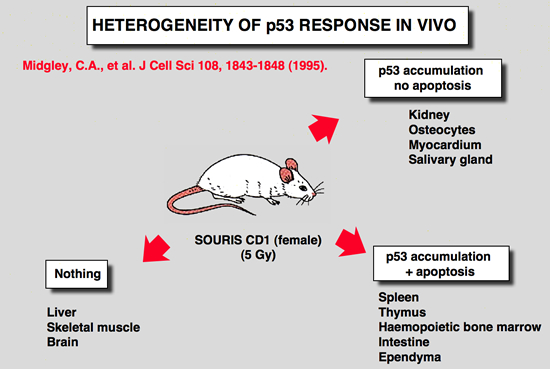
|
Midgley, C. A., B. Owens, C. V. Briscoe, D. B. Thomas, D. P. Lane and P. A. Hall (1995). “Coupling between gamma irradiation, p53 induction and the apoptotic response depends upon cell type in vivo.” J Cell Sci 108: 1843-8.
Abstract of the paper
The accumulation of p53 protein following whole body irradiation of adult mice was studied using a new polyclonal antibody to mouse p53. While dramatic accumulation of the protein was apparent in splenocytes, thymocytes and osteocytes no p53 protein accumulation was detected in the hepatocytes of the irradiated mouse. Thus, the upstream initiating signals that control the induction of p53 are controlled in a tissue specific manner. While massive apoptosis accompanies p53 induction in thymocytes and splenocytes it is not seen in the osteocytes. Thus the downstream consequences of p53 induction are also tightly controlled. These results have profound significance for an understanding of the role of the p53 tumour suppression pathway in different tissues. |
MacCallum DE, Hupp TR, Midgley CA, Stuart D, Campbell SJ, Harper A, Walsh FS, Wright EG, Balmain A, Lane DP and Hall PA (1996) The p53 response to ionising radiation in adult and developing murine tissues. Oncogene 13: 2575-2587.
Abstract of the paper
The induction of the p53 response to ionising radiation has been studied during murine development and in the adult animal. The response has been assessed by precise quantitative assay of p53 protein levels in tissues and by immunohistochemistry. Newly developed transgenic mice in which a lacZ transgene is driven by a p53 response element have also been used to directly assess the transcriptional activity of the induced protein, There is striking developmental control of the p53 response so that in early development all tissues accumulate high levels of p53 following radiation and indeed p53 is present at elevated levels in some unirradiated tissues, Later in development clear heterogeneity of the p53 response becomes apparent, both in terms of the responses of individual tissues and of cell populations, within those tissues, The study of lacZ transgene expression and the occurrence of apoptosis in different tissues that accumulate p53 protein point to a further level of control regulating the nature and degree of the downstream response to elevated levels of p53 in cells, These findings have important implications for the susceptibility of different tissue types to carcinogenic and other insults, The early expression of the p53 response is consistent with novel models of p53 function that suggest it may have evolved principally as a defense against teratogenic insult that permits plasticity of development. |
|
|






