| |
p53 Information
mouse models
ASPP family
|
p53 as an Oncogene ?
p53 as a positive cell regulator
Early work on p53 suggested that it may be implicated in the promotion of cell proliferation. Earlier experiments by Reich and Levine (Reich and Levine 1984) showed that mouse 3T3 cell growth, when arrested by serum deprivation, exhibited very low levels of p53 mRNA and protein. When the cell was induced to grow by serum stimulation, the level of p53 mRNA and the rate of p53 protein synthesis increased markedly, reaching a peak near the G1/S boundary just prior to initiation of DNA replication (Reich and Levine 1984). Similar experiments performed with normal resting T lymphocytes (Milner and McCornick 1980) and normal diploid fibroblasts (Mercer et al. 1984) showed that p53 expression is always concomitant with induction of cell growth. The level of p53 mRNA and protein is somewhat constant throughout the cell cycle when the cells are growing exponentially(Calabretta et al. 1986).
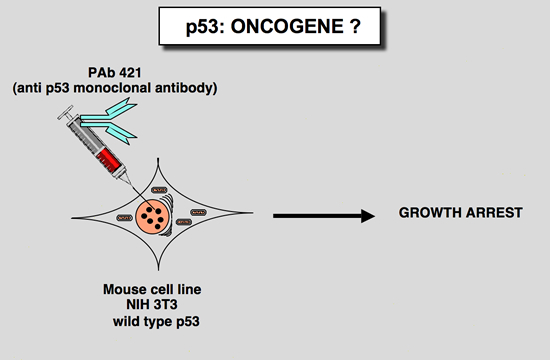
This observation, added to other characteristics of the p53 protein (short half life, nuclear localization), led to the notion that wild type p53 could play a positive role in cell proliferation. This idea was strengthed by the work of Mercer and collaborators (Mercer et al. 1984; Mercer et al. 1982). Microinjection of p53 antibody (200.47 and PAb122) into the nucleus of quiescent Swiss 3T3 mouse cells inhibited the subsequent entry of the cell into the S phase after serum stimulation. This inhibition was effective only when microinjection was performed at or around the time of growth stimulation, suggesting that p53 is critical for G0/G1 transition (Mercer et al. 1984; Mercer et al. 1982). Recently, similar results were obtained using methylcholanthrene-transformed mouse cells which express mutant p53 (Deppert et al. 1990; Steinmeyer et al. 1990). Also consistent with these results is an antisense experiment which showed that inhibition of p53 expression prevented cell proliferation in both non-transformed NIH3T3 cells and transformed cells (Shohat et al. 1987). All of these observations led to the notion that wild type p53 is a positive regulator of cell proliferation.
p53 cooperates with Ha-ras
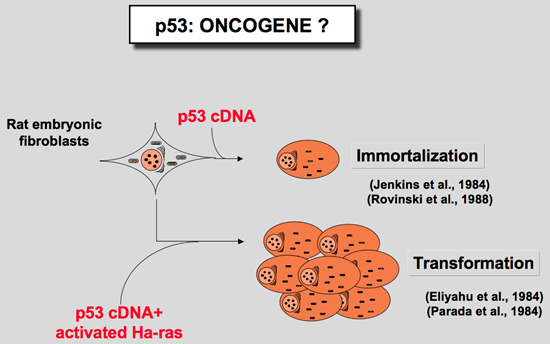
In 1984 two groups reported that cotransfection of murine p53 with plasmids encoding an activated c-Ha-ras oncogene could transform REF cells in a manner similar to that observed with proto-oncogenes such as myc or E1A (Eliyahu et al. 1984; Parada et al. 1984). A third group demonstrate that murine p53 could imortalized normal rat chondrocytes leading to cells sensitive to ras transformation (Jenkins et al. 1985; Jenkins et al. 1984). These observations resulted in the classification of p53 as a nuclear dominant oncogene.
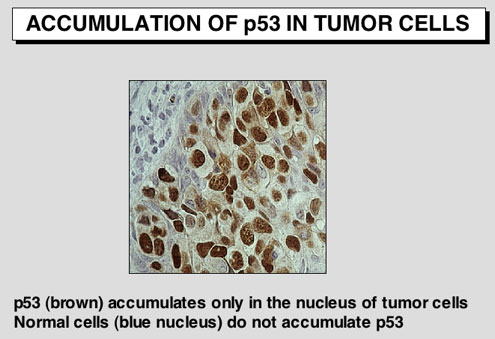
The p53 protein accumulates in murine and human tumor cells
Immunocytochemical and immunohistochemical analysis show that the p53 protein accumulates in the nucleus of transformed or tumor cells. Before 1990, the protein was believed to be wild type.
REFERENCES
• Calabretta B, Kaczmarek LL, Selleri L, Torelli G, Ming PML, Ming SC and Mercer WE (1986) Growth-dependent expression of human Mr 53,000 tumor antigen messenger RNA in normal and neoplastic cells. Cancer. Res. 46: 5738-5742.
• Deppert W, Buschhausendenker G, Patschinsky T and Steinmeyer K (1990) Cell cycle control of p53 in normal (3T3) and chemically transformed (Meth-A) mouse cells .2. requirement for cell cycle progression. Oncogene 5: 1701-1706.
• Eliyahu D, Raz A, Gruss P, Givol D and Oren M (1984) Participation of p53 cellular tumour antigen in transformation of normal embryonic cells. Nature 312: 646-649.
• Jenkins JR, Rudge K, Chumakov P and Currie GA (1985) The cellular oncogene p53 can be activated by mutagenesis. Nature 317: 816-818.
• Jenkins JR, Rudge K and Currie GA (1984) Cellular immortalization by a cDNA clone encoding the transformation-associated phosphoprotein p53. Nature 312: 651-654.
• Mercer WE, Avignolo C and Baserga R (1984) Role of the p53 protein in cell proliferation as studied by microinjection of monoclonal antibodies. Mol. Cell. Biol. 4: 276-281.
• Mercer WE, Nelson D, DeLeo AB, Old J and Baserga R (1982) Microinjection of monoclonal antibody to protein p53 inhibits serum-induced DNA synthesis in 3T3 cells. Proc Natl Acad Sci USA 79: 6309-6312.
• Milner J and McCornick F (1980) Lymphocyte stimulation: concanavalin A induces the expression of a 53k protein. Cell. Biol. Int. Rep. 4: 663-667.
• Parada LF, Land H, Weinberg RA, Wolf D and Rotter W (1984) Cooperation between gene encoding p53 tumour antigen and ras in cellular transformation. Nature 312: 649-651.
• Reich NC and Levine AJ (1984) Growth regulation of a cellular tumour antigen, p53, in non transformed cells. Nature 308: 199-201.
• Shohat O, Greenberg M, Reisman D, Oren M and Rotter V (1987) Inhibition of cell growth mediated by plasmids encoding p53 anti-sense. Oncogene 1: 277-283.
• Steinmeyer K, Maacke H and Deppert W (1990) Cell cycle control by p53 in normal (3T3) and chemically transformed (meth-A) mouse cells .1. regulation of p53 expression. Oncogene 5: 1691-1699.
|
|






