| |
p53 database
p53 database
Database curation
Download the database
Improve p53 mutation detection and report
p53 mutation and cancer
|
p53 MUTATIONS IN PROSTATE CANCER
Prostate Cancer
Carcinoma of the prostate is predominantly a tumor of older men, which frequently responds to treatment when widespread and may be cured when localized. The rate of tumor growth varies from very slow to moderately rapid, and some patients may have prolonged survival even after the cancer has metastasized to distant sites such as bone. Because the median age at diagnosis is 72 years, many patients—especially those with localized tumors—may die of other illnesses without ever having suffered significant disability from their cancer. The approach to treatment is influenced by age and coexisting medical problems. Most prostate tumors are adenocarcinomas, sharing numerous common features with other prevalent epithelial cancers, such as breast and colon cancer
A distinguishing feature of prostate cancer is its intimate association with aging; indeed, aging is the single most significant risk factor for prostate cancer. Although pre-neoplastic lesions known as prostatic intraepithelial neoplasia (PIN) can be found in men in their twenties and are fairly common in men by their fifties, clinically detectable prostate cancer is not generally manifest until the age of 60 or 70. Furthermore, the occurrence of precancerous lesions is significantly more prevalent (~1 in 3 men) than the incidence of carcinoma (~1 in 9 men). Therefore, whereas the morphological changes associated with initiation are relatively common and occur early in life, progression to invasive carcinoma is a significantly less common event that occurs in a more limited population as a consequence of aging.
Familial inheritance
Hereditary factors account for a relatively small percentage (~10%) of prostate cancers and are generally associated with early onset disease. To date, two familial susceptibility loci have been mapped to the X chromosome and to a region of chromosome 1q , although the respective candidate genes have not yet been identified. In addition, several studies have identified a statistical association between breast and prostate cancer, but the molecular basis for such a link is unresolved.
Role of steroid hormones
Steroid hormone receptor signaling plays a pivotal role in all stages of prostate carcinogenesis. In particular, there is a characteristic age-related decrease in the ratio of androgens to estrogens in men, which may represent a contributing factor in prostate cancer initiation. In addition, the transition to androgen independence that is a hallmark of advanced prostate cancer has been a focus of numerous investigations.
Heterogeneity and multifocality
The heterogeneous and multifocal nature of prostate cancer lesions poses significant difficulties for researchers. With regard to heterogeneity, histological inspection of prostate cancer tissue typically reveals a juxtaposition of benign glands, preneoplastic (PIN) foci, and neoplastic foci of varying severity. To account for this heterogeneity, Gleason proposed a grading system that is now the predominant system used by pathologists, since it is an excellent prognostic indicator. In this system, a score is given based on the sum of the two most prevalent grades of neoplastic foci (e.g., 3 + 3; 3 + 4); a higher Gleason grade indicates a more advanced carcinoma.
p53 and Prostate cancer
Loss of chromosome 17p occurs in advanced stages of prostate cancer and metastatic disease, deleting a region that includes the p53 locus, but not BRCA. It is now generally accepted that mutations of p53 occur infrequently in early invasive carcinoma (Henke et al. 1994; Voeller et al. 1994; Prendergast et al. 1996), although not all studies concur (Chi et al. 1994). In contrast, p53 is mutated in advanced stages of prostate cancer, as well as in recurrent and metastatic disease, as inferred by immunocytochemical localization of accumulated protein (presumed to be indicative of its mutation) or by direct mutational analysis (Bookstein et al. 1993; Effert et al. 1993; Navone et al. 1993; Aprikian et al. 1994; Eastham et al. 1995; Heidenberg et al. 1995), again with dissenting reports (Dinjens et al. 1994). Moreover, several studies indicate that p53 overexpression is a predictive factor for poor prognosis and disease recurrence, particularly when detected in combination with Bcl2 (Thomas et al. 1993; Shurbaji et al. 1995; Bauer et al. 1996; Moul et al. 1996; Matsushima et al. 1997; Theodorescu et al. 1997; Brewster et al. 1999; Stackhouse et al. 1999).
Interestingly, the frequency of p53 mutations seems to be lower in prostate cancer than in other cancers. A relatively minor role for p53 in prostate carcinogenesis is consistent with the observation that Li-Fraumeni patients carrying germline p53 mutations have a low incidence of prostate cancer (Kleihues et al. 1997), although it may be the case that they succumb to other carcinomas before they can develop prostate cancer. An alternative possibility is that the variability in reported p53 mutation rates reflects a differential tissue-specific role of other components in the p53 pathway, particularly mdm2 and p21 (Osman et al. 1999). A final possibility is that p53 mutations occur frequently in prostate cancer, but are difficult to detect, since they appear to be associated with more highly aggressive and/or recurrent forms of the disease, for which tissue acquisition is more difficult (Visakorpi et al. 1992; Yang et al. 1996).
Spectrum of p53 mutations in prostate cancer
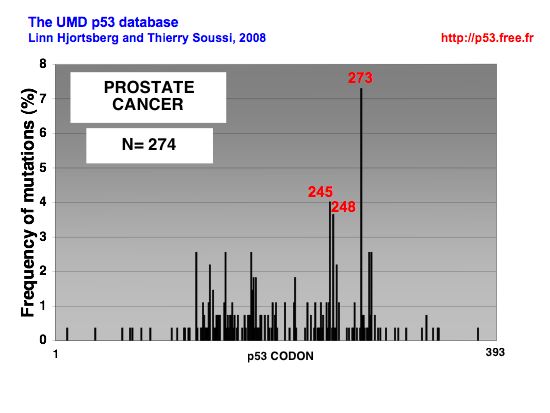 |
DISTRIBUTION OF p53 MUTATIONS IN PROSTATE CANCER |
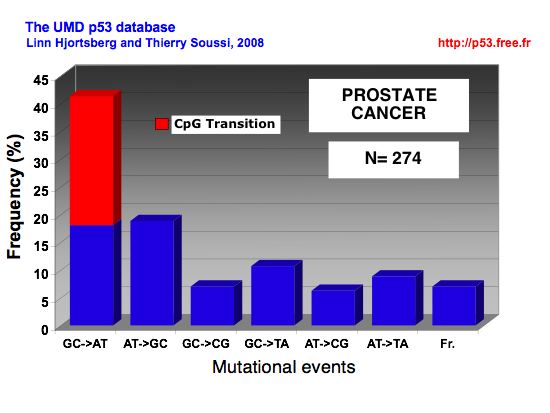 |
MUTATIONAL EVENTS IN PROSTATE CANCER |
|
Meta-analysis of p53 loss of function in prostate cancer.
Points ; mean p53 activity as measured by transactivation with the WAF1promoter ; bars , 95 % CI. The mean and 95 % CI of p53 activity for all studies combined for a specific type of cancer is shown on the far left of each graph.
Horizontal line, mean of the combined studies. The publication code is indicated on the x-axis : the first number is an anonymous ID for the publication and the second number indicates the number of p53 mutants included in this study. Studies are presented from left to right in decreasing order of number of p53 mutants. The y-axis corresponds to p53 transactivation activity, with a value of 1.5 for the negative control and a value of 2.5 for 100 % of wild-type activity. Only studies with 5 or more p53 mutations are shown on the graph.
More information about this statistical analysis can be found in this article:
Soussi, T., Asselain, B., Hamroun, D., Kato, S., Ishioka, C., Claustres, M. and Beroud, C. (2006) Meta-analysis of the p53 mutation database for mutant p53 biological activity reveals a methodologic bias in mutation detection. Clin Cancer Res, 12, 62-69. Download the pdf
|
|
|






